Volunteer Water Sampling with KELT
KELT started volunteer water sampling in the Kennebec Estuary in 2013 in the town of Georgetown. In 2014, sampling expanded to include Phippsburg. Now, there are 19 sites sampled across the 2 towns. We've also partnered with other groups that test coastal water nearby to create the Maine Coastal Observing Alliance. Through this partnership, we have been able to collect profiles of the estuary waters from a boat at 5 sites since 2014. In 2018, we partnered with the Maine Department of Marine Resources to start sampling phytoplankton at one site in Georgetown.
How are the waters tested?
- WAKE Volunteer Manual: All the details about our methods and procedures for water sampling are included in our volunteer manual.
- WAKE Quality Assurance Project Plan: Our guidelines for maintaining data quality are outlined in our Quality Assurance Project Plan, approved by the Maine DEP in 2016.
What Are We Testing and Why?
Weather Conditions
Why Record the Weather?
Weather has the potential to cause variations in the conditions we observe while sampling. We might see slightly low dissolved oxygen conditions but realize that the day was overcast. Maybe one day the salinity will show up a bit low, but we can use the weather information to tie it to a large rainfall.
The weather conditions that you record at your site form the foundation of all your other sampling day tests.
The weather conditions we are testing include air temperature, wind direction, wind speed (based on the Beaufort scale), % cloud cover, and precipitation.
Temperature
Why Test Temperature?
Temperature is an important characteristic to test for both its direct and indirect impacts on marine life.
On their own, temperature measurements can give valuable information about the conditions at a sample site. Understanding the rate that water warms up in the spring and summer and cools down in the fall can help to define characteristics of an area. Organisms are adapted to live at certain ranges of temperature at certain times of year and can be stressed when the temperature goes outside of its typical range. Eggs and juvenile fish are particularly sensitive to temperature. Temperature measurements can also reveal potential impacts from fresh water sources or offshore sources of water when tied in with salinity information.
Temperature’s relationship to pH, dissolved oxygen, water density, and the metabolic rates of organisms makes it essential that temperature is tested at the same time as any of these other characteristics.
Testing Temperature
Water temperature will be tested using the old tried and true method; a glass thermometer. These thermometers measure temperature in degrees Celsius. The thermometers can measure temperatures between -5°C (23°F) and 55°C (122°F). The liquid inside the thermometer is non-toxic.
Water Clarity
Why Measure Water Clarity?
Water clarity, also called Turbidity, has a large impact on organisms living in the water. The more clear the water, the more sunlight can penetrate the water and reach plants under the surface. The more sunlight plants have access to, the more they can photosynthesize and produce oxygen, raising the amount of DO in the water.
There are two main sources of the cloudiness we might observe in the water: suspended sediment or algae. Large amounts of suspended sediment are frequently found during windy weather or after a storm. Large amounts of algae may indicate that there are sources of nutrients in the area that are causing the plants to grow very well. The nutrients may come from fertilizers or animal waste. Our nitrate/nitrite/ammonia measurements we help to determine if these sources of nutrients are present.
Testing for Water Clarity
A Secchi disk will be used to measure water clarity. This will only be measured at sites where there is a dock because deep water is needed for the Secchi disk method. The Secchi disk is dropped into the water, and the depth where the disk is no longer visible is noted. This depth indicates the degree of water clarity.
Salinity
What Can Salinity Measurements Tell Us?
In Georgetown, a town impacted by both fresh water, from rivers and from the land’s surface, and salt water, from the ocean, salinity measurements are key to identifying where that water is coming from. Water with low salinity suggests that there is a large impact from the Kennebec River, Sheepscot River, and town of Georgetown. High salinity water suggests that the impact is from offshore and tidal areas. As we test other characteristics, including pH, DO, and Nitrate/Nitrite/Ammonia, salinity measurements can help us to understand the sources of differences we may note.
Testing for Salinity
Salinity will be tested using a hydrometer. Because salinity is a very stable water characteristic, samples will be collected in the field in 1 L bottles and brought back to the Georgetown Town Hall for analysis.
Dissolved Oxygen
What is Dissolved Oxygen?
Dissolved oxygen (DO) is all of the oxygen gas that is floating around in the water. It is essential for marine fish, mammals, plants, and other organisms to breathe. If the concentration of oxygen drops to between 3 to 5 mg/L, marine organisms are stressed. If the amount of oxygen drops below 3 mg/L, fish and other marine organisms can die.
What Factors Impact the Amount of Dissolved Oxygen?
Dissolved oxygen enters the water through three primary methods: exchange with the air along the water’s surface, the release of oxygen through photosynthesis of marine plants, algae and phytoplankton, and by splashing and waves that mix air with the water.
The temperature of the water, salinity, water clarity, and the amount of organisms using the oxygen can change the amount of DO in the water. Cold water has the ability to hold more gasses than warm water. That is why cold soda is fizzier than warm soda. High water temperatures can result in low DO levels. Salinity has a similar relationship to DO. The higher the salinity, the less oxygen the water is able to hold.
Impacts of Salinity and Temperature on the Amount of Dissolved Oxygen that Water Can Hold
Water clarity (turbidity) relates to the amount of dissolved oxygen due to the impact it has on plants and algae. Clear water allows in the most sunlight, so plants are able to complete more photosynthesis and release more oxygen into the water. Less clear water blocks the sunlight, limiting the amount that plants can photosynthesize.
Organisms can also use up the oxygen in the water. This process is called eutrophication. It typically happens when there is an overpopulation of certain organisms and then a large die off of those organisms. Examples of this include a large fish kill or excessive amounts of algae that die off at the end of the season. These dead organisms then begin to rot. This means that there are millions of bacteria living and breathing as they break down the remains of these organisms. The bacteria use large amounts of oxygen, resulting in unhealthy, low DO levels.
Because of the factors that impact dissolved oxygen, it changes throughout the day. Plants only photosynthesize during the day, so just before dawn dissolved oxygen levels are typically low. No photosynthesis has been happening all night, but organisms have still been breathing and using the oxygen. In the middle of the day, when the sun’s rays are shining most directly on the plants and algae, the most oxygen is being produced.
In warm summer weather, the high amounts of oxygen in the middle of the day are tempered by the high temperatures in the middle of the day. The resulting increases in water temperature decrease the amount of available oxygen.
Testing Dissolved Oxygen
We will be testing dissolved oxygen using the Winkler Titration method.
This Method is a two part process. The first part ‘fixes’ the amount of oxygen in the water, preventing it from escaping or being added from the surrounding air so that the amount you measure is accurate for your sample site. To fix the oxygen, two solutions are added. They contain Mn+2 ions and OH– ions that bond to form a white compound that settles to the bottom of the sample. All the dissolved oxygen in the sample reacts with this white compound to form new compounds that are brown in color. The oxygen is then trapped in those compounds and can’t escape into the air. A strong acid is then added that dissolves all of the compounds. At this point, the amount of oxygen in the sample is stable. As the compounds dissolve and form new chemicals, iodine (I2) is released, two iodine molecules for each dissolved oxygen molecule that was in the water.
The second part of this process measures the amount of iodine (and the corresponding amount of oxygen). The iodine will have turned the sample a yellow/brown color. Into a measured amount of the treated sample (25mL), sodium thiosulfate () is added in measured drops. The sodium thiosulfate bonds with the iodine to form new compounds, two sodium thiosulfates used for each iodine. This removes the iodine from solution and the yellow/brown color lightens. Before the iodine color disappears, a starch is added that is blue when iodine is in the sample, to make the color change easier to observe. When all the iodine is gone, the blue color will disappear. Because you will have measured how much sodium thiosulfate you used to make the iodine disappear, you will be able to figure out exactly how much oxygen was present in the water. The units on the titrator that is used to add the sodium thiosulfate are set up so that 1 unit on the titrator equals 1mg/L of dissolved oxygen.
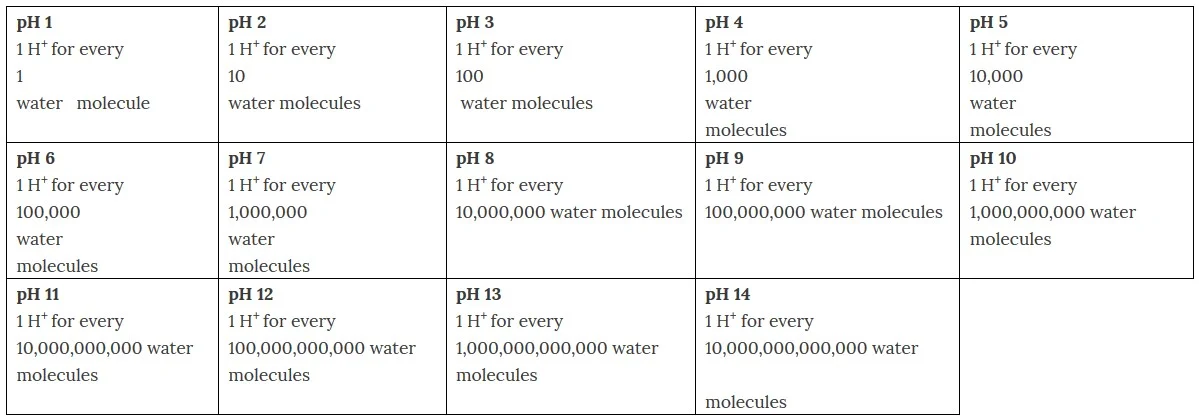
pH
What is pH?
pH is a measurement of acidity, ranging on a scale from 1 to 14. Less than 7 is considered acidic, more than 7 is considered basic, and 7 is considered neutral.
What makes something acidic, basic, or neutral?
- The charged particles (ions) in the solution. The two key ions are H+ and OH–.
- When there is more H+, the solution is acidic (<7 on the pH scale).
- When there is more OH–, the solution is basic (>7 on the pH scale).
- Pure water, H2O, has an even number of H+ and OH– ions, so it is considered neutral and has a pH of 7.
Water found in nature, in streams, rain, groundwater, or the ocean, rarely has a pH of exactly 7. Gasses from the air, minerals from soil and rocks, or chemical inputs from human practices mix with the water and result in the water having either more H+ ions or more OH– ions.
pH in the World Around Us
Gasses like carbon dioxide (CO2) or nitrogen dioxide (NO2) cause the pH to decrease when they mix with water. The natural carbon dioxide in the atmosphere causes the average pH of rain water to be 5.6. As the amount of carbon dioxide in the atmosphere increases, this number will decrease. Acid rain, with a pH of 4 or less, occurs in areas where there are a large amount nitrogen dioxide and sulfur dioxide (SO2) emissions, from power plants or car exhaust, that mix with the rain water.
Other compounds that have broken down from minerals in rocks can have the opposite impact, either increasing or helping to stabilize the pH. Two ions that are found in large amounts in ocean water are carbonate (CO3-2) and borate (H2BO3–). These ions that have a negative charge, so they can bond with positively charged hydrogen ions to form new compounds. This removes H+ from solution and raises the pH (decreasing the acidity). They also bond with any new H+ ions that are added to the water. This serves to buffer the water pH, so the concentration of H+ stays constant, and the pH doesn’t change.
Because of these compounds, the average pH of the ocean is fairly consistent throughout, ranging from 8.0 to 8.3. Ocean life is adapted to live at that pH level. Fresh water lakes usually have more variable and typically lower pH than the ocean because there are fewer mineral ions to buffer the water. Lakes and streams typically have pH values ranging from 6.5 to 8.0.
Carbonate plays another key role in the ocean. Many types of ocean life, from tiny single celled creatures to giant corals, shellfish, and some types of algae, take carbonate out of the water to make their shells. The carbonate bonds with calcium ions (Ca+2) to form calcium carbonate (CaCO3). In water with a low pH (high acidity), too much of the carbonate is already bonded to H+, so there is not enough available for animals to make shells. If the pH drops too low, delicate calcium carbonate shells can begin to dissolve. The pH and the amount of available carbonate in the water only need to drop low enough that the water does not have an over-saturation of carbonate. Recent studies have shown that this can begin to happen at a pH of 7.5 in the ocean. Decreases in pH have the potential to harm Maine’s shellfish populations.
Global and Local Changes in Ocean pH
The pH of the ocean as a whole is decreasing. This process, called ocean acidification, is happening because there are increasing amounts of CO2 in the atmosphere. This CO2 mixes with water in the ocean and in rain to form carbonic acid. Because there is such a large influx of CO2 to the atmosphere, the ocean is unable to completely buffer this added acid. As a result, the pH of the oceans has decreased globally by 0.1 pH units, a 30% increase in H+ concentration, over the past 100 years.
Although the ocean as a whole is not anywhere near the 7.5 level, there are several reasons that we may find pH values lower than the global average in the waters around Georgetown. Because fresh water typically has a lower pH than ocean water, the mixing of Sheepscot and Kennebec River waters with ocean water in the estuary around Georgetown can lead to localized areas with lower pH values. Another potential reason the pH might be low is that inputs of nutrients from the surface of the land, from fertilizers and organic waste, can lead to blooms of algae. When these blooms die and rot, the rotting releases organic acids that can locally decrease the pH of an area. Areas with large amounts of rotting organic material from dead plants or animals also have high amounts of organic acids released during the decay of the organisms that decrease the pH.
Georgetown’s Shellfish Committee has expressed an interest in finding out more about the pH of Georgetown’s waters. Clam populations seem to be decreasing in some areas of town, and no one has yet identified the reason for those decreases.
Testing pH
We will be testing pH using Baker pH paper. This pH paper has a fine resolution so that we will be able to identify relatively small changes in pH. Using three sets of pH paper, we will be able to test with a detail that ranges from 0.1 to 0.3 pH units within the range of pH 6.0 to pH 10.0.
The pH scale is not an arbitrary set of numbers; it is a precise measurement of the concentration of H+ ions in solution. The smaller the number, the higher the amount of H+ ions. The pH scale is a negative log scale, so each number smaller on the pH scale has 10 times as many H+ ions in solution. The change of even a single pH unit means a large change in the concentration of H+ ions and a large change in the acidity. A change of 0.1 on the pH scale can mean that the amount of H+ ions has doubled.
Phytoplankton
Our phytoplankton sampling program will support the DMR's harmful algal bloom monitoring program. This program tests for the microscopic algae that live in the water and cause problems like red tide.
Click here to find out more information about the DMR's program